Oh, the weather outside if frightful…
But the fire is so delightful.
And since we’ve no place to go
I’ll tell you, how it is, there is snow!
Lame attempt at musical humor aside, did you ever wonder where all that white stuff actually comes from?
Its magic!
Mother Nature waves her magic wand &….
Really though, it seems easy…water vapor up in the air cools off & voila…snow!!!
But here’s the rest of the story…
First you may need a refresher course in water.
- Water molecules – H2O – are made up of hydrogen & oxygen.
- In the liquid state the molecules resemble Mickey Mouse’s head.
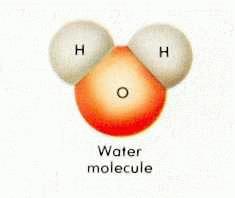 You may remember this graphic from the previous post on the water cycle…(Dinosaur Pee Sept 8th)
You may remember this graphic from the previous post on the water cycle…(Dinosaur Pee Sept 8th)
- Hydrogen atoms are negatively charged & repel each other when they get close. Remember elementary science lessons with magnets? – opposite magnetic poles attract & likes repel
- In the liquid state, water molecules move about quickly & that movement is stronger than the repelling force, so the molecules can get relatively close together.
- When the water is cooled, the movement of the molecules slows down & (here’s the tricky part so pay attention!) the hydrogen atoms are able to better repel each other, which means they are pushed farther apart.
Here’s what happens way up in the sky…
- Lovely water molecules float around up in the air & some of them join together to form droplets.
- Some droplets get pushed up by wind currents.
- When its colder in the upper part of the sky the molecules in the droplets slow down.
- When they reach about 4 degrees Celsius they begin to spread out.
- In addition to the lovely water molecules floating around in the sky, there is microscopic debris, mainly in the form of dust, dirt and salt. This debris can come from anywhere in the world, carried by the wind. It gives droplets something to cling to. When you catch a snowflake on your tongue, you could be eating a speck of sand from the Mojave, soil from the Andes Mountains, or even dust from under your own bed.
- Now you have these cold droplets floating around, getting colder, & when they near the freezing point ( 0 degrees Celsius) they begin for form crystals.
- The crystals attract other crystals which begin to form snowflakes.
- Wind, temperature, & the amount of water droplets in the air all work together to determine the size, shape, & number of snowflakes.
- Mean old Mr. Gravity does the rest of the work. When the snowflakes are heavy enough, he pulls them down to us.
That’s the science of snow.
Here’s the magic…
All snowflake pictures are courtesy of SnowCrystals.com
And since we’ve no place to go, Let it snow, let it snow, let it snow!













Beautiful thoughts